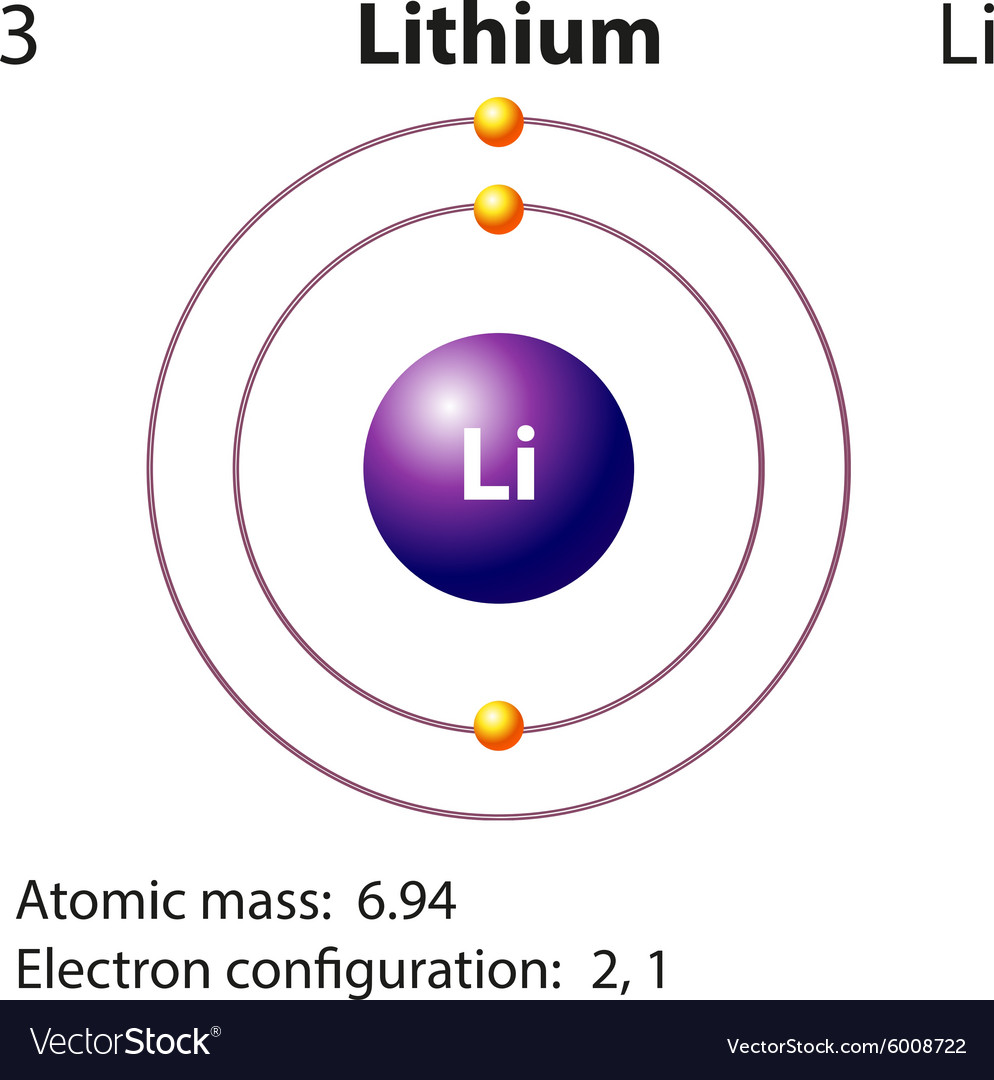
Compound 3 was not catalytically active toward the borylation reaction, but it showed excellent catalytic performance in the hydroboration of aldehydes and ketones by pinacol borane (HBpin), leading to a series of boric esters with high yield. The borylation reactions feature a wide substrate scope, high functional group versatility and mild conditions. Compounds 1 and 4 were both active for this coupling reaction, and complex 4 displayed much higher activity than that of 1. The catalytic activities of 1–4 toward the borylation of aryl iodides with 4,4,4′,4′,5,5,5′,5′-octamethyl-2,2′-bis(1,3,2-dioxa-borolane) (B 2Pin 2) were studied. In compound 4 Tol, only one β-diketimine unit was deprotonated, and the two zinc atoms were bridged by a chloride atom. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. A molecule consists of two or more atoms joined. Compound 3 reacted with anhydrous zinc chloride in a stoichiometric ratio of 1 : 2 to produce Element Lithium (Li), Group 1, Atomic Number 3, s-block, Mass 6.94. A lithium atom has a still heavier nucleus and three electrons. The deprotonation of bis(β-diketiminato) ligand H 2L 2 by nBuLi generated complex ( 3). The reactions of ZnEt 2 with one equivalent of and two equivalents of ligand HL 1, which bears a large 2,4,6-tricyclopentylphenyl substituent, afforded compounds ( 1) and ( 2), respectively. Hydrogen atoms all have one electron occupying the space outside of the nucleus. Since atoms are neutral, the number of electrons in an atom is equal to the number of protons. The metal itselfwhich is soft, white, and lustrousand several of its alloys and compounds are produced on an industrial scale. Neutron Number and Mass Number of Lithium Mass numbers of typical isotopes of Lithium are 6 7. 1: The social security number subatomic-the proton. lithium (Li), chemical element of Group 1 (Ia) in the periodic table, the alkali metal group, lightest of the solid elements. Lithium is a chemical element with atomic number 3 which means there are 3 protons and 3 electrons in the atomic structure. K87 Li II Ground State 1s 2 1 S 0 Ionization energy 610078 cm-1 (75.6400 eV) Ref. The syntheses, characterizations, and catalytic activities of three zinc complexes and one lithium compound based on β-diketiminate ligands are described. Lithium atoms have three protons, beryllium atoms have four, and so on. Li I Ground State 1s 2 2s 2 S 1 / 2 Ionization energy 43487.150 cm-1 (5.391719 eV) Ref.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
